Formula:
Amount of Substance = Mass (m) / Molar mass
(M)
Amount of Substance Calculator
An Amount of Substance Calculator is a tool used to calculate the amount of substance in moles. The amount of substance is a measure of how much of a given substance is present, typically expressed in moles. It is commonly used in chemistry to understand the quantity of particles (atoms, molecules, or ions) in a sample.
Why use an Amount of Substance Calculator?
- Stoichiometric calculations: It is essential for determining how much reactant or product is involved in a chemical reaction, which is useful for balancing equations and predicting outcomes.
- Laboratory work: It helps scientists and students quickly calculate the number of moles when preparing solutions, mixing chemicals, or analyzing compounds.
- Chemical analysis: In both research and industrial applications, it's used to quantify how much of a substance is present in a sample.
How does an Amount of Substance Calculator work?
The amount of substance, measured in moles (mol), is calculated using the following formula:
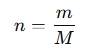
where:
- n is the amount of substance (in moles).
- m is the mass of the substance (in grams).
- M is the molar mass of the substance (in grams per mole, g/mol).
Example:
If you have 10 grams of water (H2O), the molar mass of water is approximately 18 g/mol. The amount of substance (in moles) would be:
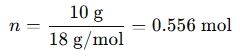
Some calculators might also work with:
- Volume: If you're working with gases at standard conditions, you can calculate the number of moles based on volume using molar volume (for gases) at specific conditions.
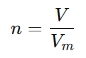
where V is the volume and Vm is the molar volume (e.g., 22.4 L/mol for ideal gases at STP).
When to use an Amount of Substance Calculator?
- Chemical reactions: When performing stoichiometric calculations to determine the proportions of reactants and products in a reaction.
- Preparing solutions: When you need to know how many moles of a solute are required to prepare a specific concentration of a solution.
- Quantifying elements and compounds: To calculate how many moles of a substance are in a sample or how many molecules are present.
- Gas calculations: When dealing with gases and needing to calculate the number of moles from volume (at standard temperature and pressure).