Formula:
N = (Z * M)(D * V)
Avogadro's Number Calculator
What is an Avogadro's Number Calculator?
An Avogadro's Number Calculator is a tool used to work with Avogadro’s constant, 6.022×1023, which represents the number of atoms, molecules, or particles in one mole of a substance. This number is a fundamental constant in chemistry and physics.
The calculator helps convert between:
- Moles of a substance
- Number of particles (atoms, molecules, ions)
- Mass of a substance (when used alongside molar mass)
Why use an Avogadro's Number Calculator?
- Counting tiny particles: Atoms and molecules are incredibly small, so counting them directly isn’t practical — Avogadro’s number makes this possible.
- Converting between moles and particles: Chemistry often requires switching between macroscopic measurements (like grams) and microscopic counts (like atoms).
- Balancing chemical equations: Knowing how many particles react or form in a reaction.
- Determining molecular quantities: Calculating the amount of substance in chemical reactions and solutions.
How does an Avogadro's Number Calculator work?
- Converting moles to particles:
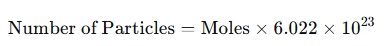
- Converting particles to moles:
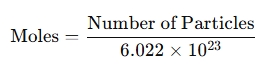
- Working with mass: When combined with the molar mass of a substance:
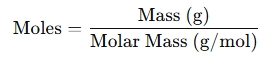
- Combining conversions: To find the number of particles from mass:
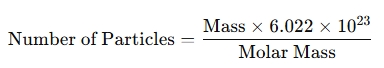
When is an Avogadro's Number Calculator used?
- In chemistry labs: For measuring and preparing chemical solutions.
- In physics: When studying gases and the behavior of particles.
- In stoichiometry: Balancing equations and predicting product amounts.
- In material science: Calculating atomic-scale properties from bulk measurements.
- In education: Teaching and learning the mole concept and atomic theory.