Formula:
PiVi / Ti = PfVf / Tf
Combined Gas Law Calculator
What is a Combined Gas Law Calculator?
A Combined Gas Law Calculator calculates the relationship between pressure, volume, and temperature of a gas when the amount of gas (in moles) is constant. It combines Boyle's Law, Charles's Law, and Gay-Lussac's Law into one equation:
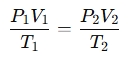
Where:
- P1 , P2 = Initial and final pressures
- V1 , V2 = Initial and final volumes
- T1 , T2 = Initial and final temperatures (in Kelvin)
Why use a Combined Gas Law Calculator?
- Simplify Calculations: It combines multiple gas laws into one equation, making it easier to solve problems without needing to use separate formulas.
- Real-World Applications: Helps analyze and predict the behavior of gases in various conditions, such as in engines, weather systems, or industrial processes.
- Efficiency: Quickly calculates changes in pressure, volume, or temperature when other values are known.
How does a Combined Gas Law Calculator work?
- Input: You provide the initial and final values for pressure, volume, and temperature (usually in SI units: pressure in pascals, volume in cubic meters, temperature in Kelvin).
- Process: The calculator uses the combined gas law equation to solve for the unknown variable (pressure, volume, or temperature).
- Output: The result gives you the new value of the unknown property based on the initial conditions.
Example:
If you know the initial pressure P1=2 atm, volume V1=5 L, and temperature T1=300 K, and you need to find the final pressure P2 when V2=10 L and T2=350 K, you can input these into the formula and calculate P2 .
When should you use a Combined Gas Law Calculator?
- Gas-related Problems: When you need to find how pressure, volume, or temperature changes for a gas in a closed system, such as in physics or chemistry problems.
- Real-World Applications: In engineering and environmental science, when dealing with gases under varying conditions (e.g., in engines, refrigeration systems, or weather modeling).
- Laboratory Work: In labs where precise control of gas conditions is necessary.