Equivalent Weight Calculator
What is an Equivalent Weight Calculator?
An Equivalent Weight Calculator is a tool used to calculate the equivalent weight of a substance, which is the mass of the substance that reacts with or supplies one mole of a given substance in a chemical reaction. In simpler terms, the equivalent weight is the mass of a substance that will combine with or displace a fixed amount of another substance in a reaction.
The equivalent weight depends on the valency of the substance (how many electrons or ions it can donate or accept) and its molar mass. For an acid, base, or salt, the formula for calculating equivalent weight is:
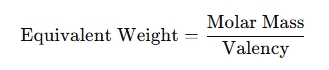
For example:
- In the case of sulfuric acid (H2SO4), the molar mass is 98 g/mol, and the valency of H2SO4 (since it can donate 2 protons) is 2. So, the equivalent weight is:
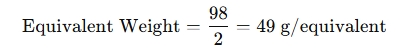
Why use an Equivalent Weight Calculator?
- Accurate chemical calculations: Helps in determining the right amount of a substance to use in reactions.
- Acid-base titrations: Used to calculate how much acid or base is required to neutralize a solution.
- Stoichiometric calculations: For finding the correct proportions of reactants and products in chemical reactions.
- Concentration calculations: Helps find the concentration of a solution in terms of equivalent weight.
- Electrochemical applications: Used in electrolysis and redox reactions to determine the amount of substance involved in an oxidation-reduction process.
How does an Equivalent Weight Calculator work?
- Input the substance: Enter the name or formula of the substance whose equivalent weight is needed.
- Determine the molar mass: The calculator finds the molecular weight (molar mass) of the substance (in g/mol).
- Input the valency or equivalence factor: The valency depends on the substance type. For acids, it refers to the number of protons it can donate. For bases, it refers to the number of hydroxide ions it can donate or accept.
- Apply the formula: It uses the formula:
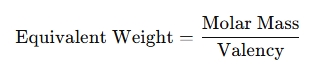
- Output the equivalent weight: The result will be the equivalent weight of the substance in grams per equivalent.
For example, if you're working with sulfuric acid:
- Molar mass = 98 g/mol
- Valency = 2 (since H2SO4 can donate 2 protons)
The equivalent weight would be:
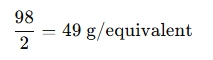
When is an Equivalent Weight Calculator used?
- In chemistry experiments: To determine the quantities of reactants needed for specific reactions.
- In titration processes: Especially in acid-base titrations, where you need to know how many equivalents of acid or base are present.
- In manufacturing: For industries like pharmaceuticals and chemicals, where precise amounts of substances need to be combined or reacted.
- In electrochemistry: When determining the amount of substance involved in electrolysis or redox reactions.
- In education: To understand and calculate equivalents for various chemical reactions.