Equivalent Mass of an Acid Calculator
What is an Equivalent Mass of an Acid Calculator?
An Equivalent Mass of an Acid Calculator is a tool that determines the equivalent mass of an acid, which is the mass of the acid that donates one mole of hydrogen ions (H⁺) in a reaction. It helps in titration calculations and chemical analysis.
The formula to calculate the equivalent mass of an acid is:
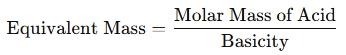
where basicity is the number of replaceable hydrogen ions (H⁺) per molecule of the acid.
Why use an Equivalent Mass of an Acid Calculator?
- Quick and accurate calculations for lab experiments.
- Essential in titration to determine normality and equivalent weight.
- Helps in stoichiometry for balancing acid-base reactions.
- Useful in industrial applications like pharmaceuticals and food chemistry.
How does it work?
- Input the molar mass of the acid (from the periodic table).
- Enter the basicity (number of H⁺ ions the acid donates).
- The calculator divides the molar mass by basicity to give the equivalent mass.
Example Calculation:
For Sulfuric Acid (H₂SO₄)
- Molar Mass = 98 g/mol
- Basicity = 2 (because it donates 2 H⁺)
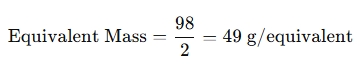
When to use this calculator?
- In titration experiments to determine acid strength.
- In chemistry labs for normality and stoichiometry.
- For industrial and pharmaceutical applications involving acids.
- In educational settings for students learning acid-base chemistry.